MP-1000

Compact, lightweight system enables easy attachment during training!
MP-1000 makes it possible to offer more appropriate risk management and post-discharge exercise guidance during rehabilitation through its continuous,high-precision management of vital data.
Characteristics








Bluetooth connection for smartphone monitoring

Enables monitoring of exercise intensity and efficiency that couldn't previously be evaluated.
MP-1000 a set that enables easy attachment without the need for cables or electrodes stuck to the chest. Attached to the patient during training it continuously measures blood oxygen saturation and the pulse from the earlobe. The device has made it possible to quantitatively evaluate the intensity and efficiency of exercise based on recorded vital data. It can be used to support decisions on the allowable level of exercise and lifestyle guidance after discharge during rehabilitation.
Also supports home-based rehabilitation.Collectively managing data in the cloud.
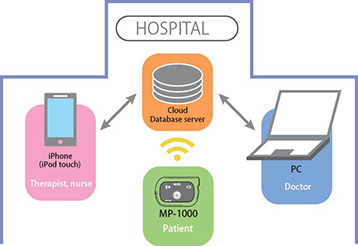 The MP-1000's measurement data is collected on a cloud server in a system accessible on PCs and smartphones at the time of monitoring. Data files may be displayed in each mode enabling measurement results to be viewed in more detail. This makes it possible to manage rehabilitation at hospitals and offer appropriate care for patients who are undergoing rehabilitation at home.
The MP-1000's measurement data is collected on a cloud server in a system accessible on PCs and smartphones at the time of monitoring. Data files may be displayed in each mode enabling measurement results to be viewed in more detail. This makes it possible to manage rehabilitation at hospitals and offer appropriate care for patients who are undergoing rehabilitation at home.
Measurement stability during exercise
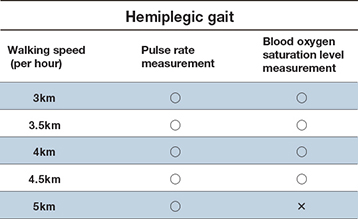 The accuracy of measurements for treadmill walking by hemiplegia patients has been verified for speeds of up to 5 km/hour for pulse measurements and 4.5 km/hour for blood oxygen saturation readings(varies depending on the individual).
The accuracy of measurements for treadmill walking by hemiplegia patients has been verified for speeds of up to 5 km/hour for pulse measurements and 4.5 km/hour for blood oxygen saturation readings(varies depending on the individual).
Meets ISO standards established in 2011
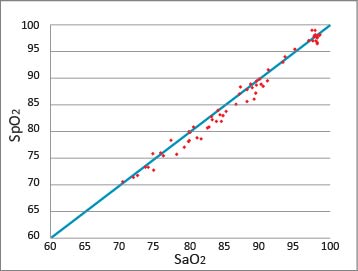 The ISO standard (ISO 80601-2-61:2011) criteria require a system to be tested on humans. Pulse Oximeter MP-1000 was confirmed to meet the standards in an"evaluation test of SpO2 precision levels on humans" conducted in a low-oxygen chamber at the Travelers medical Center, Tokyo Medical University Hospital.
The ISO standard (ISO 80601-2-61:2011) criteria require a system to be tested on humans. Pulse Oximeter MP-1000 was confirmed to meet the standards in an"evaluation test of SpO2 precision levels on humans" conducted in a low-oxygen chamber at the Travelers medical Center, Tokyo Medical University Hospital.
The graph is an excerpt from the article, "ARE ALL PULSE OXIMETERS RELIABLE AT SEVERE HYPOXIC CONDITION?" (Authors: Nao Kurita, Shigeru Masuyama, Shinji Fukushima, and Atsuo Hamada. Travelers medical Center, Tokyo Medical University Hospital, Tokyo Japan) released at the 19th International Hypoxia Symposium held between March 3-8, 2015.
MP-1000 SPEC
- Model
- MP-1000
- Measurement principle
- Double wavelength light absorption method
- Measurement location
- Ear lobe / fingertip
- LCD
- -
- Blood pressure measurements
- -
- Measurement of pulse rate
- ○
- Display of pulse amplitude strength
- -
- Event measurement
- -
- Display of perfusion index value
- -
- Out of range warning
- -
- Energy conservation mode
- -
- Automatic switching of direction
- -
- Memory
- Memory in iOS device
- Finger circumference
- -
- Bluetooth
- Bluetooth Low Energy 4.1
- Power supply
- Lithium ion battery
- Battery life (continuous use)
- approx. 8 hours
- Dimensions (H x W x D)
- 47 x 71.5 x 20mm
- Weight
- approx. 40g(without batteries)
- Water resistance
- IP22





